Lab 6d Testing Plant Substances as Potential Medicines
Purpose: We will find what plant materials, that are native, contains elements that will help bacteria grow.
Materials:
Balance, weigh boat, lab scoops
LB broth base
Media bottles, 250 mL
Sterilize/autoclave
Water bath, 37 degrees Celsius, shaking
Sterile LB agar
Laminar flow hood and disinfectant
Glasses, safety, plastic
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Petri dishes, 60 x 15mm, sterile
E.Coli JM109 (stock plate)
Plant specimen
Mortar and pestle
Pipet, 10 mL and pump
Plastic funnels, short-stemmed
Filter paper disks, 5 mm diameter
Beakers, 100 mL
Syringe, 10 mL and filter, .2 microliters
Reaction tubes and rack, 1.7 mL
Methanol, absolute
Pipet, 1 mL and pump
Dry block heater/ heat block
Forceps, fine-tipped
Ampicillin
Glass spreader
Incubator oven, 37 degrees Celsius
Procedure
1. Get mortar and pestle and grind 2 grams of plant with 10 mL of di H20. Then let it sit for 3 minutes. After this let this paste filter through a piece of paper that is lined with a funnel. Let this filter sterilize (.2 micrometers using syringe) and put it in a 1 mL microtube.
2. Repeat step 1, but instead of distilled water use 10 mL of Methanol. It is very toxic so use gloves and goggles.Put this extract in a heat block and leave the lid open to evaporate and leave it overnight. The next day add 1 mL of sterile water and vortex.
3. Sterilize forceps by putting it in flames, then put 2 discs in each of the extract and leave it overnight for the H20 extract or 15 minutes for the Methanol extract.
4. Once again get sterilized forceps and put 2 discs in each microbe with H20 (negative control).
5. Use flame to sterilize forceps once last time and put 1 disc in microtube with ampicillin (positive control).
6. Sterilize 1 mL pipet and transfer 1 mL of E. coli culture to a petri dish. Before you do that part divide the petri dish into 4 quadrants. Then spread the E. coli into all four quadrants with a sterilized glass spreader. Then cover the petri dish and let it soak for 15 minutes.
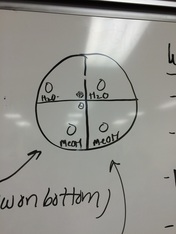
7. Sterilize forceps and place discs on the plates in each of the four quadrants s shown on the diagram below.
8. Incubate this plate at 37 degrees Celsius for 24 hours.
Materials:
Balance, weigh boat, lab scoops
LB broth base
Media bottles, 250 mL
Sterilize/autoclave
Water bath, 37 degrees Celsius, shaking
Sterile LB agar
Laminar flow hood and disinfectant
Glasses, safety, plastic
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Petri dishes, 60 x 15mm, sterile
E.Coli JM109 (stock plate)
Plant specimen
Mortar and pestle
Pipet, 10 mL and pump
Plastic funnels, short-stemmed
Filter paper disks, 5 mm diameter
Beakers, 100 mL
Syringe, 10 mL and filter, .2 microliters
Reaction tubes and rack, 1.7 mL
Methanol, absolute
Pipet, 1 mL and pump
Dry block heater/ heat block
Forceps, fine-tipped
Ampicillin
Glass spreader
Incubator oven, 37 degrees Celsius
Procedure
1. Get mortar and pestle and grind 2 grams of plant with 10 mL of di H20. Then let it sit for 3 minutes. After this let this paste filter through a piece of paper that is lined with a funnel. Let this filter sterilize (.2 micrometers using syringe) and put it in a 1 mL microtube.
2. Repeat step 1, but instead of distilled water use 10 mL of Methanol. It is very toxic so use gloves and goggles.Put this extract in a heat block and leave the lid open to evaporate and leave it overnight. The next day add 1 mL of sterile water and vortex.
3. Sterilize forceps by putting it in flames, then put 2 discs in each of the extract and leave it overnight for the H20 extract or 15 minutes for the Methanol extract.
4. Once again get sterilized forceps and put 2 discs in each microbe with H20 (negative control).
5. Use flame to sterilize forceps once last time and put 1 disc in microtube with ampicillin (positive control).
6. Sterilize 1 mL pipet and transfer 1 mL of E. coli culture to a petri dish. Before you do that part divide the petri dish into 4 quadrants. Then spread the E. coli into all four quadrants with a sterilized glass spreader. Then cover the petri dish and let it soak for 15 minutes.
7. Sterilize forceps and place discs on the plates in each of the four quadrants s shown on the diagram below.
8. Incubate this plate at 37 degrees Celsius for 24 hours.
9. Measure qualitative and quantitative data. Personally my results were negative for inhibiting bacteria growth. The bacteria lawn around each disk was about 2mm. Around the positive disk (ampicilin) the clearing was about 3mm, while the negative control (distilled water) was about 2mm. Both of my MeOH tested negative because they both fostered bacteria growth. For my H20 it also tested negative because bacteria also grew next to the disc. The reason it was negative because bacteria grew around the disc and there was a clearing of no bacteria.
Data Analysis/ Conclusion
My extracts didn't give me positive results because there was bacteria was growing around the disc and the whole point of the lab was to break down the bacteria. My controls did give me the results as I expected because the negative control had bacteria growing around it while the positive control had a clearing of bacteria. Errors that could give me false results is that I contaminated the discs in the positive and negative results. Also I may have forgot to sterilize my forceps after each time I stuck them in my tubes to extract the discs. The next steps I could do is repeat this lab and that time use more discs for the MeOH and H20. Maybe that time I could use three to see if the majority either passed for being positive or negative.
Thinking Like a Biotechnician
1. No it doesn't mean that the extract is microbial agent because there are many error that could be committed. But if the lab was conducted perfectly then yes the extract isn't a microbial agent because bacteria grew (which is what by meaning it has a negative result).
2. If the methanol extractions smell too much like alcohol then when conducting the experiment you wouldn't know if the molecules killed the bacteria or if the alcohol itself did.
3. We would use some type of chromatography to extract each individual molecule to identify the exact compound.
Data Analysis/ Conclusion
My extracts didn't give me positive results because there was bacteria was growing around the disc and the whole point of the lab was to break down the bacteria. My controls did give me the results as I expected because the negative control had bacteria growing around it while the positive control had a clearing of bacteria. Errors that could give me false results is that I contaminated the discs in the positive and negative results. Also I may have forgot to sterilize my forceps after each time I stuck them in my tubes to extract the discs. The next steps I could do is repeat this lab and that time use more discs for the MeOH and H20. Maybe that time I could use three to see if the majority either passed for being positive or negative.
Thinking Like a Biotechnician
1. No it doesn't mean that the extract is microbial agent because there are many error that could be committed. But if the lab was conducted perfectly then yes the extract isn't a microbial agent because bacteria grew (which is what by meaning it has a negative result).
2. If the methanol extractions smell too much like alcohol then when conducting the experiment you wouldn't know if the molecules killed the bacteria or if the alcohol itself did.
3. We would use some type of chromatography to extract each individual molecule to identify the exact compound.